|
Kids in diapers aren’t allowed in the foam pit. There is also a climbing wall, climbing ropes and a trampoline. 
It has a set of trapeze swings that the kids had a great time swinging from to land in the foam pit. My little guy had a great time driving his own kart and running into the inflatable dividers.įoam Pit – We also spent plenty of time in the foam pit. There’s a height limit on this one, so smaller kids can’t drive the big karts, but they do have a Go Karts downstairs for younger kids. Go Karts – Indoor Go Karts are located on the uppermost floor. My 5 year-old was especially loved driving his own car. I actually lost count of how many times we went on them. The course was filled with pirates, mythical creatures and other fun things to see.īumper Cars – My kids loved these. Mini Golf – It's 18 holes of black light mini-golf, and I loved the way the black lights made everything from the balls to the holes and wall decorations glow. Here are the activities we enjoyed most at Rush Funplex. Here's what you can expect at The Rush Funplex. 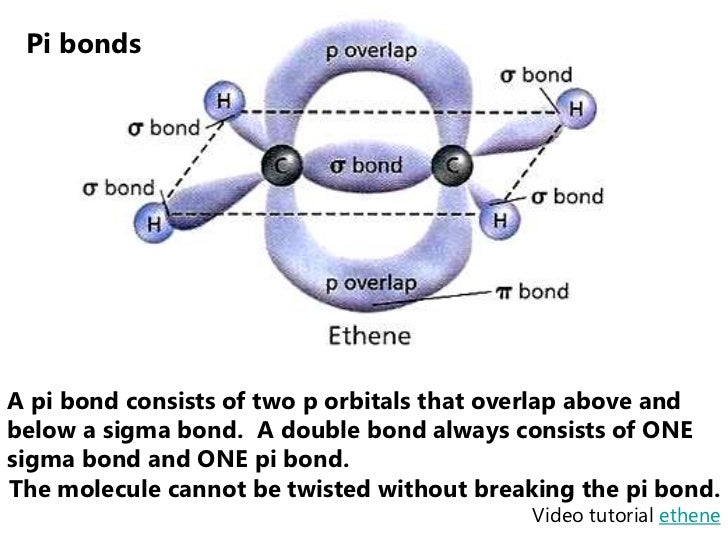
Everything is inside, so it doesn’t matter if it’s hot, snowy, cold or rainy. We had a blast! There are so many things to do. I had a great coupon for The Rush Funplex, so I ventured out with a couple of our kids, ages 13 and 5. 
I’m always looking for things that I can do with our kids near our home. It was a hit with both my teen and my 5-year-old. This rule fails further when considering other shapes - toroidal fullerenes will obey the rule that the number of sigma bonds in a molecule is exactly the number of atoms plus the number of rings, as will nanotubes - which, when drawn flat as if looking through one from the end, will have a face in the middle, corresponding to the far end of the nanotube, which is not a ring, and a face corresponding to the outside.Need a fun all-ages, all-weather activity in Davis County, Utah? Our family discovered The Rush Funplex in Syracuse last summer. Ordinarily, one extra face is assigned to the space not inside any ring, but when Buckminsterfullerene is drawn flat without any crossings, one of the rings makes up the outer pentagon the inside of that ring is the outside of the graph. This is because the sigma rule is a special case of the Euler characteristic, where each ring is considered a face, each sigma bond is an edge, and each atom is a vertex. This rule fails in the case of molecules which, when drawn flat on paper, have a different number of rings than the molecule actually has - for example, Buckminsterfullerene, C 60, which has 32 rings, 60 atoms, and 90 sigma bonds, one for each pair of bonded atoms however, 60 + 32 - 1 = 91, not 90. In this case there are 16 C−C sigma bonds and 10 C−H bonds. The anthracene molecule, C 14H 10, has three rings so that the rule gives the number of sigma bonds as 24 + 3 − 1 = 26. 
Molecules with rings have additional sigma bonds, such as benzene rings, which have 6 C−C sigma bonds within the ring for 6 carbon atoms. There is no more than 1 sigma bond between any two atoms. This rule is a special-case application of the Euler characteristic of the graph which represents the molecule.Ī molecule with no rings can be represented as a tree with a number of bonds equal to the number of atoms minus one (as in dihydrogen, H 2, with only one sigma bond, or ammonia, NH 3, with 3 sigma bonds). According to the sigma bond rule, the number of sigma bonds in a molecule is equivalent to the number of atoms plus the number of rings minus one. Organic molecules are often cyclic compounds containing one or more rings, such as benzene, and are often made up of many sigma bonds along with pi bonds. These sigma bonds can be supplemented with other bonding interactions, such as π-back donation, as in the case of W(CO) 3( PCy 3) 2(H 2), and even δ-bonds, as in the case of chromium(II) acetate. Transition metal complexes that feature multiple bonds, such as the dihydrogen complex, have sigma bonds between the multiple bonded atoms. For example, propane is described as consisting of ten sigma bonds, one each for the two C−C bonds and one each for the eight C−H bonds. The concept of sigma bonding is extended to describe bonding interactions involving overlap of a single lobe of one orbital with a single lobe of another. Sigma bonds are obtained by head-on overlapping of atomic orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
